Hyperbaric Oxygen Therapy (HBOT) activates neuroplasticity in chronic stroke patients by reawakening dormant neurons in the ischemic penumbra. A 2013 randomized controlled trial showed that 40 HBOT sessions at 2.0 ATA (atmospheres absolute) produced measurable increases in brain metabolic activity on SPECT imaging, corresponding to significant improvements in neurological function and daily living. This article explains the science behind those findings and what they mean for stroke survivors.
What Is Neuroplasticity and Why Does It Matter After Stroke?
Neuroplasticity is the brain’s ability to reorganize itself by forming new neural connections. After a stroke, the brain does not simply accept the damage. Surviving neurons can take over functions previously handled by damaged areas. New synapses form. Existing neural pathways strengthen.
This process is most active in the first weeks and months after stroke, which is why early rehabilitation is so important. But neuroplasticity does not stop entirely after that window closes. The question is whether it can be meaningfully reactivated later, and whether HBOT can serve as a catalyst for that reactivation.
The Ischemic Penumbra: Where HBOT Does Its Work
During a stroke, a core area of brain tissue dies due to total blood flow loss. Surrounding that dead core is a zone called the ischemic penumbra. These are neurons that survived the stroke but were damaged by oxygen deprivation. They remain structurally intact but metabolically suppressed. They are alive, but they are not functioning.
In the acute phase, the penumbra is the target of emergency interventions like tPA and thrombectomy, which aim to restore blood flow before these cells die. But in many patients, penumbral tissue persists for months or years in a state of metabolic dormancy.
This is the tissue that HBOT targets. By delivering 100% medical-grade oxygen at elevated pressure (typically 2.0 ATA), HBOT dramatically increases the amount of dissolved oxygen in the blood plasma. This oxygen reaches areas where red blood cells may struggle to deliver oxygen through damaged microvasculature. The result is that dormant penumbral neurons receive the oxygen they need to resume function.
The Efrati 2013 Trial: Proof That Late Neuroplasticity Is Real
The most important study on HBOT and stroke neuroplasticity is the 2013 prospective randomized crossover trial by Efrati et al., published in PLoS ONE. It enrolled 74 patients (59 completed) who had suffered strokes 6 to 36 months earlier. All had completed standard rehabilitation and were no longer improving.
Patients received 40 sessions of HBOT at 2.0 ATA, 90 minutes per session, five days per week. The crossover design was critical: half the patients received HBOT immediately, while the other half served as controls for two months before crossing over to receive treatment.
The Clinical Results
Patients who received HBOT showed significant improvements in NIHSS (National Institutes of Health Stroke Scale), activities of daily living, and quality of life. Patients in the control period showed no improvement. When control patients crossed over and received HBOT, they also improved.
This crossover design is powerful because it demonstrates that the improvements were attributable to HBOT, not to spontaneous recovery or the passage of time. Patients who were stable for months suddenly improved once treatment began.
The Imaging Evidence
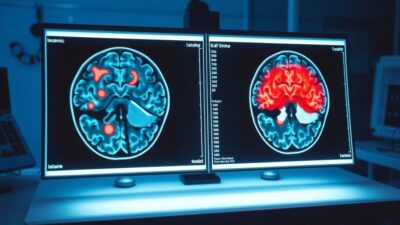
SPECT (single-photon emission computed tomography) brain imaging provided the mechanistic explanation. Before HBOT, scans showed regions where anatomical structure was intact but metabolic activity was low, the hallmark of the dormant penumbra. After treatment, these same regions showed significantly increased metabolic activity.
In other words, HBOT was reactivating neurons that had been alive but dormant for months. This is late neuroplasticity: the brain rewiring itself well past the conventional recovery window.
How HBOT Triggers Neuroplasticity: The Biological Mechanisms
HBOT promotes neuroplasticity through multiple interconnected pathways. These are not theoretical. They are documented in both brain health research and stroke-specific studies.
Oxygen-Driven Gene Expression
The cycling of high-pressure oxygen (during sessions) and normal pressure (between sessions) creates a pattern known as the hyperoxic-hypoxic paradox. This triggers the expression of hypoxia-inducible factor 1 (HIF-1) and vascular endothelial growth factor (VEGF), both of which promote the formation of new blood vessels (angiogenesis) and neural repair.
A 2021 comprehensive review by Gottfried et al., published in Biomolecules, documented these mechanisms and their role in cognitive improvement after HBOT. The review, cited by over 100 subsequent papers, established that HBOT activates transcription factors including HIF-1, VEGF, and SIRT-1.
Angiogenesis: New Blood Vessels to Starved Tissue
One of the most important mechanisms is angiogenesis. HBOT stimulates the growth of new blood vessels into areas of the brain with compromised circulation. A 2015 study by Tal et al. demonstrated that HBOT induced cerebral angiogenesis in post-traumatic brain injury patients, with perfusion MRI confirming increased cerebral blood flow and volume even a decade after the initial injury.
In stroke patients, this is particularly relevant. The penumbra suffers from damaged microvasculature. New blood vessel growth restores oxygen delivery to these regions, supporting sustained neural function even after the HBOT course ends.
Reduced Neuroinflammation
Chronic inflammation in the brain suppresses neural function and inhibits plasticity. HBOT has been shown to reduce neuroinflammation by modulating inflammatory cytokines and reducing oxidative stress. The Rosario et al. 2018 study of 7 ischemic stroke patients documented changes in serum biomarkers for inflammation and neural recovery after 40 HBOT sessions, alongside improvements in cognition, executive function, and gait.
Enhanced Mitochondrial Function
Mitochondria are the energy producers of every cell, including neurons. After stroke, mitochondrial dysfunction in the penumbral zone contributes to ongoing neural suppression. HBOT provides the raw material (oxygen) for mitochondrial ATP production, helping cells resume normal metabolic function. Animal studies have shown that HBOT upregulates brain-derived neurotrophic factor (BDNF) and activates autophagy via the AMPK-mTOR pathway, clearing damaged cellular components and supporting new growth.
What “Brain Rewiring” Actually Means for Daily Function
The neuroscience is compelling, but what matters to stroke survivors is function. What does neuroplasticity look like in practice?
The Hadanny et al. 2020 study of 162 chronic stroke patients provides the most detailed picture. After 40 to 60 HBOT sessions, 86% achieved clinically significant cognitive improvement across multiple domains:
- Memory: Better ability to retain and recall information
- Attention: Improved ability to focus and sustain concentration
- Information processing speed: Faster response times and mental processing (hemorrhagic stroke patients showed the greatest gains here)
- Executive function: Better planning, decision-making, and problem-solving
The Khairy et al. 2025 case report illustrates the motor side. A 45-year-old man with hemorrhagic stroke, 15 months post-event, progressed from wheelchair dependence to walking with a cane after 83 HBOT sessions. DTI imaging showed increased structural connectivity in white matter tracts, and SPECT confirmed a 15.83% increase in motor cortex perfusion.
These improvements represent the brain literally reorganizing: dormant neurons reactivating, new synaptic connections forming, and new blood vessels supplying areas that had been oxygen-starved.
HBOT as a Catalyst, Not a Replacement for Rehabilitation
An important framing: HBOT does not replace physical therapy, occupational therapy, or speech therapy. It amplifies them. The neuroplasticity triggered by HBOT creates a window of enhanced brain adaptability. Rehabilitation exercises performed during this window may produce greater gains because the brain is more receptive to learning and adaptation.
Think of it this way. Rehabilitation provides the repetition and task-specific practice that drives neural reorganization. HBOT provides the biological conditions (oxygen, blood flow, reduced inflammation) that make the brain more responsive to that practice. The combination may be more effective than either approach alone, though direct head-to-head trials of combined protocols are still limited.
For more on combining these approaches, see our article on HBOT for recovery.
Cognitive Recovery vs. Motor Recovery
The current evidence shows a stronger and more consistent signal for cognitive improvement than for motor improvement after HBOT in chronic stroke patients. The Hadanny 2020 data showed significant gains across all cognitive domains. Motor improvements are documented (the Khairy case report is one example, and the Efrati 2013 trial showed ADL improvements), but the effect appears less dramatic in most patients.
This may be because cognitive function relies more on diffuse neural networks that can be reactivated across the penumbra, while motor recovery often depends on specific tracts (like the corticospinal tract) that may be more severely damaged. The distinction is important for setting realistic expectations.
Who Is Most Likely to Respond?
Based on the available research, the following factors appear to predict better outcomes:
- Identifiable penumbral tissue: Patients whose brain imaging shows regions with structural integrity but low metabolic activity are the best candidates. This mismatch indicates recoverable tissue.
- Higher baseline cognitive function: The Hadanny 2020 study found that baseline cognitive function was the strongest predictor of improvement across all domains.
- Both stroke types respond: There was no significant difference between cortical and subcortical strokes. Hemorrhagic stroke patients showed a particular advantage in information processing speed.
- Time since stroke is less important than expected: Benefits have been documented in patients from 6 months to nearly 16 years post-stroke.
fMRI and SPECT Evidence: Watching the Brain Change
One of the most compelling aspects of HBOT for stroke neuroplasticity is that we can see the changes happening in real time with brain imaging. Two types of imaging provide the most useful data.
SPECT Imaging
SPECT (single-photon emission computed tomography) measures regional cerebral blood flow and metabolic activity. In the Efrati 2013 study, pre-treatment SPECT scans identified brain regions where anatomical structure was intact (visible on CT) but metabolic activity was suppressed. These are the dormant penumbral neurons.
After 40 HBOT sessions, post-treatment SPECT showed significant increases in metabolic activity in these same regions. The spatial correlation between the imaging changes and the clinical improvements provided strong evidence that HBOT was not simply producing a placebo effect, but was driving real biological changes in specific brain regions.
In the Khairy 2025 case, SPECT quantified the changes precisely: a 15.83% increase in right motor cortex perfusion and a 15.92% increase in right frontal lobe perfusion. These numbers corresponded directly to the patient’s motor improvement (wheelchair to cane) and cognitive gains (improved attention and memory).
Diffusion Tensor Imaging (DTI)
DTI is an MRI technique that maps white matter tracts, the “wiring” that connects different brain regions. After stroke, damaged white matter tracts show reduced fractional anisotropy (a measure of structural integrity). The Khairy 2025 case showed that after HBOT, fractional anisotropy increased in major white matter tracts, indicating that structural connectivity was improving.
This is particularly significant because white matter repair is a hallmark of neuroplasticity. It means the brain was not just reactivating existing neurons, but strengthening the connections between them. This is what functional rewiring looks like on imaging.
Perfusion MRI
The Tal et al. 2015 study used whole-brain perfusion analysis to demonstrate that HBOT induced cerebral angiogenesis (new blood vessel growth) in post-brain-injury patients. Both cerebral blood flow and cerebral blood volume increased significantly, even in patients who were more than a decade past their initial injury.
For stroke patients, perfusion MRI can serve as both a diagnostic tool (identifying recoverable tissue before treatment) and an outcome measure (confirming that blood flow has improved after treatment). This dual role makes it increasingly important in HBOT research protocols.
How Long Do the Neuroplasticity Effects Last?
A critical question for any neuroplasticity intervention: do the gains persist, or do they fade once treatment stops?
The Rosario 2018 study provides the most direct answer for HBOT in stroke. Functional improvements in cognition, executive function, and gait were maintained at 3 months post-treatment. However, serum biomarkers for inflammation and neural recovery returned toward baseline more quickly than the functional improvements did.
This suggests that the structural and functional changes induced by HBOT (new synaptic connections, reactivated neurons, new blood vessels) may outlast the acute biochemical effects. Once neurons are reactivated and synaptic connections are strengthened, they may remain functional even without ongoing HBOT, particularly if the patient continues active rehabilitation.
Longer-term follow-up data (beyond 6 months post-treatment) is limited. Some clinicians recommend periodic “booster” sessions (5 to 10 sessions every 3 to 6 months) to maintain and build on initial gains, though this approach has not been validated in controlled studies.
The durability question is one of the most important gaps in the current evidence base. Future studies with 12-month and 24-month follow-up assessments will be essential for understanding the long-term trajectory of HBOT-induced neuroplasticity in stroke patients.
Neuroplasticity in Younger vs. Older Stroke Patients
Age is often assumed to be a barrier to neuroplasticity. Older brains are generally considered less plastic than younger brains. However, the HBOT stroke data does not clearly support this assumption.
In the Hadanny 2020 study, the mean age was 60.75 years, with patients ranging across a wide age spectrum. Age was not reported as a significant predictor of treatment response. Baseline cognitive function, not age, was the strongest predictor of improvement.
The Hadanny/Efrati 2020 RCT on healthy aging (separate from the stroke study) demonstrated that HBOT produced large effect sizes for attention (0.745) and information processing speed (0.788) in adults over 64. MRI confirmed increased cerebral blood flow in key brain regions. This suggests that the aging brain retains sufficient plasticity to respond to HBOT stimulation.
For stroke patients, the implication is that advanced age should not be an automatic disqualifier. A 70-year-old with viable penumbral tissue and good baseline cognition may respond as well as a 50-year-old. Individual assessment matters more than age alone.
Limitations and What We Still Do Not Know
The neuroplasticity story is promising but incomplete. Several limitations deserve attention:
- Most positive data comes from the Efrati/Hadanny research group at the Sagol Center in Israel. Independent replication from other centers with sham-controlled designs is essential.
- The 86% improvement rate is from a retrospective single-center study, not a multi-site RCT.
- We do not yet know the optimal timing to start HBOT, the minimum number of sessions needed, or whether booster sessions are required to maintain gains.
- HBOT for stroke is not FDA-cleared and is considered investigational.
- Long-term durability of improvements beyond 3 to 6 months post-treatment is not well studied in large cohorts.
For a broader look at HBOT for brain conditions, including traumatic brain injury and cognitive decline, see our dedicated hub page.
The Bottom Line on HBOT and Stroke Neuroplasticity
The evidence supports a compelling biological story: HBOT reactivates dormant neurons, promotes new blood vessel growth, reduces neuroinflammation, and creates conditions for the brain to rewire itself. In chronic stroke patients who have plateaued with conventional rehabilitation, 40 or more sessions at 2.0 ATA have produced meaningful improvements in cognitive function, with brain imaging confirming the underlying biological changes.
This is not a cure. It is a tool that, for the right patient, may extend the window of recovery well beyond what was previously thought possible. For full details on the evidence for HBOT in stroke recovery, see our comprehensive guide.
Sources
- Efrati S, Fishlev G, Bechor Y, et al. “Hyperbaric Oxygen Induces Late Neuroplasticity in Post Stroke Patients – Randomized, Prospective Trial.” PLoS ONE. 2013;8(1):e53716. DOI: 10.1371/journal.pone.0053716
- Hadanny A, et al. “Hyperbaric oxygen therapy improves neurocognitive functions of post-stroke patients.” Restorative Neurology and Neuroscience. 2020;38(1):93-108.
- Khairy S, et al. “Anatomical and metabolic brain imaging correlation of neurological improvements following HBOT.” Journal of Medical Case Reports. 2025;19:87. DOI: 10.1186/s13256-025-05577-5
- Gottfried I, Schottlender N, Ashery U. “Hyperbaric Oxygen Treatment – From Mechanisms to Cognitive Improvement.” Biomolecules. 2021;11(10):1520. DOI: 10.3390/biom11101520
- Tal S, Hadanny A, Berkovitz N, et al. “HBOT may induce angiogenesis in patients suffering from prolonged post-concussion syndrome.” Restorative Neurology and Neuroscience. 2015;33(6):943-951. DOI: 10.3233/RNN-150585
- Rosario ER, et al. “The Effect of HBOT on Functional Impairments Caused by Ischemic Stroke.” Neurology Research International. 2018;2018:3172679. DOI: 10.1155/2018/3172679
- Hadanny A, et al. “Cognitive enhancement of healthy older adults using hyperbaric oxygen: a randomized controlled trial.” Aging. 2020;12(13):13740-13761. DOI: 10.18632/aging.103571
- Wang S, et al. “Enriched oxygen improves age-related cognitive impairment through enhancing autophagy.” Frontiers in Aging Neuroscience. 2024;16:1340117. DOI: 10.3389/fnagi.2024.1340117
Medical Disclaimer
The content on BaricBoost.com is for informational purposes only and is not intended as a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.